REVIEW ARTICLE
Magnes Res 1989 Sep;2(3):195-203
Recommended dietary amounts of magnesium: Mg RDA
J. Durlach
CHU Cochin--Port Royal, 27 Rue du Fg St. Jacques, F-75674
Paris Cédex 14, France
Preliminary data on this subject were presented at the
International Workshop "Recommended Dietary Amounts of Minerals"
organized by the International Life Sciences Institute, European
Branch, at Limelette Castle near Brussels, May 5-6, 1989.
Summary: In developed countries,
the recommended dietary amounts of magnesium have been set at 6
mg/kg day. The magnesium requirements for optimal health in the
adult population depend on mesological and constitutional
conditioning factors. They may intervene at every stage of
magnesium metabolism: absorption, circulation, storage and
excretion. The influence of other nutrients is more significant
on magnesium absorption than on urinary excretion. Among the
multiple interactions it is important to emphasize the
maintenance of a Ca/Mg ratio close to 2 in the intake. Magnesium
deficit and stress reinforce each other in a pathogenic vicious
circle. The Bw35 allele of HLA typing and behavioural
type A discriminate two constitutional factors increasing
magnesium requirements. The effective passive regulatory
mechanism for magnesium overload, the lability of the active
regulatory mechanisms for magnesium deficit and the considerable
need for exchangeable magnesium are factors which attribute
special importance to balance studies in determining the
magnesium intake which prevents negative magnesium balance and
magnesium deficiency.
Marginal primary magnesium deficit affects a large
proportion of the population (15 to 20%), in keeping with a daily
mean magnesium intake slightly over 4 mg/kg day versus
the Mg RDA of 6 mg/kg day. A physiological oral magnesium load
test, evaluated through non-specific and specific clinical and
paraclinical items, constitutes the best proof that the clinical
pattern depends on an insufficient magnesium intake, confirmed
after one month of supplementation. Further research appears
necessary. It would be advisable to carry out long-term magnesium
balance studies in European countries on the self-selected diets
of adults, together with comparisons of direct evaluation of
magnesium in the diet with data obtained from tables of food
composition. Magnesium intervention trials should be planned to
determine whether magnesium supplements decrease the pathogenic
consequences of magnesium deficit and particularly to evaluate
the efficiency of magnesium supplements in latent tetany
(hyperventilation syndrome, "idiopathic" Barlow's disease).
Supplementation should consist of a high magnesium density
nutrient such as magnesium in water, which has better
bioavailability than magnesium-fortified foods.
Key words: Balance, deficit,
excess intake, load, magnesium, marginality, needs, recommended
allowances, requirements, supplementation, turnover.
Introduction
Magnesium is an essential metal. The magnesium requirement
should not be determined in terms of an absolute amount. The
requirement is relative since it is strictly correlated with
various mesological and constitutional conditioning factors.
Therefore recommended dietary amounts of magnesium (Mg RDA) must
agree with the data from balance studies in healthy adults and
with prevention of the clinical manifestations of marginal
primary magnesium deficit.
The aim of this paper is (1) To analyse the conditioning
factors of magnesium requirements; (2) To define Mg RDA in
developed countries according to balance studies; (3) To show the
consistency between Mg RDA and prevalence of marginal primary
magnesium deficit in human beings; and (4) To suggest further
research.
Conditioning factors of Mg requirements
I shall successively consider magnesium essentiality and the
related physiological oral magnesium load test, the concepts of
absolute and relative requirements and the nature of the
conditioning factors.
Mg essentiality and physiological oral Mg load test
In 1926, Jehan Leroy proved the essential character of
magnesium for mice. Subsequently, in the thirties the groups of
E. MacCollum and D.M. Greenberg revealed many of the
physiological properties of magnesium, examining in the rat the
multiple effects of a lack of magnesium in the diet on
development, reproduction, neuromuscular apparatus and humoral
balance. They then studied the specific reversibility of such
deficits using a physiological oral load of magnesium. These
studies provided the first experimental basis in animals for a
diagnostic test of magnesium deficiency by oral loading with
physiological doses of magnesium, and while high intakes of
magnesium have non-specific pharmacological effects, oral
physiological doses are devoid of any such activity. Their
effectiveness in a clinical context constitutes the best tool to
diagnose magnesium deficiency1.
Absolute and relative requirements
"Absolute magnesium requirement" is an erroneous concept for
magnesium--as well as for all the minerals. In experimental
deficiency, it is possible to study the effects of the lack of a
single nutrient in the same environmental conditions and in a
homogeneous strain. Such an approach does not mirror the
multiplicity of interacting factors that affect nutritional
requirements. The magnesium requirement is thus relative,
according to individual differences which may be either genetic
or environmental1,2.
Conditioning factors of Mg requirements
Conditioning factors may intervene at every stage of magnesium
metabolism: absorption, circulation, storage and excretion.
Intestinal absorption is the sum of simple passive diffusion (one
third) and of active process of facilitated diffusion (two
thirds). The latter may play a role in regulating processes.
Magnesaemia is remarkably stable in healthy subjects. On the
average less than 30% of the Mg intake is absorbed. Total body Mg
is 1000 mmol, i.e. 24 g. The maintenance of the gradient
between intra- and extra-cellular Mg is active: efflux by active
diffusion and influx by facilitated diffusion, which are both
energy-dependent processes.
Excretion is essentially renal. It is very important to note
that reabsorption functions very close to saturation1.
Intestinal excretion and sweat are generally of secondary
importance.
Mesological conditioning factors
The main mesological conditioning factors which concern Mg
requirements are other constituents of the diet and stress among
the environmental factors.
Other nutrients and Mg requirement
All the macronutrients and micronutrients interfere with Mg
requirements. Slightly acid diets rich in protein (especially
animal protein), unsaturated fatty acids, medium chain
triglycerides and volatile fatty acids, sodium, vitamin B,
lactose and vitamin D favour Mg absorption. The absorption of
magnesium is also linked to that of water, which modifies the
physicochemical nature of magnesium1-3. Lastly, diets
which enhance the secretion of insulin, PTH, 1,25(OH2)D and
perhaps certain polypeptide digestive hormones (i.e. VIP, CEP)
improve Mg absorption and thus may also intervene in Mg storage.
Conversely some vegetable proteins, ammonia compounds, saturated
fats, dietary fibres, phytic acid, an excess of phosphorus,
calcium or alcohol, and slight alkalinity tend to inhibit Mg
absorption1.
Expansion of extracellular fluid (especially in cases of
sodium sensitivity), any agent causing hypercalcaemia (calcium,
vitamin D, lactose), proteins, all sugars metabolized in the
kidney (such as glucose), alcohol, acidifiers, diets which
enhance ADH, and aldosterone all increase magnesuria.
Conversely phosphorus and the D vitamins (in moderate doses)
and diets which enhance secretion of calcitonin, PTH, the
insulin, or glucagon reduce magnesuria.
The influence of other nutrients on Mg requirement has a more
significant effect on Mg absorption than on urinary Mg
excretion4. Among the multiple interactions between
various nutrients and magnesium, it is particularly important to
stress the maintenance of a Ca/Mg ratio close to 2 in the
intake5.
Stress and Mg requirement
Stress is capable of inducing magnesium deficit through two
main types of neurohormonal mechanisms. Stress makes three major
regulatory systems of Mg homeostasis ineffective: it replaces
hypersecretion of physiological doses of adrenaline by large
amounts of catecholamines, it reduces secretion of insulin, and
it increases urinary loss of taurine. Stress induces three types
of hypersecretion (ADH, thyroid hormones and corticoids) which
increase magnesium urinary leakage. These different elements
combine to induce a state of magnesium depletion that is due
mainly to hypermagnesuria (Fig. 1, Stress induction of magnesium
deficit).
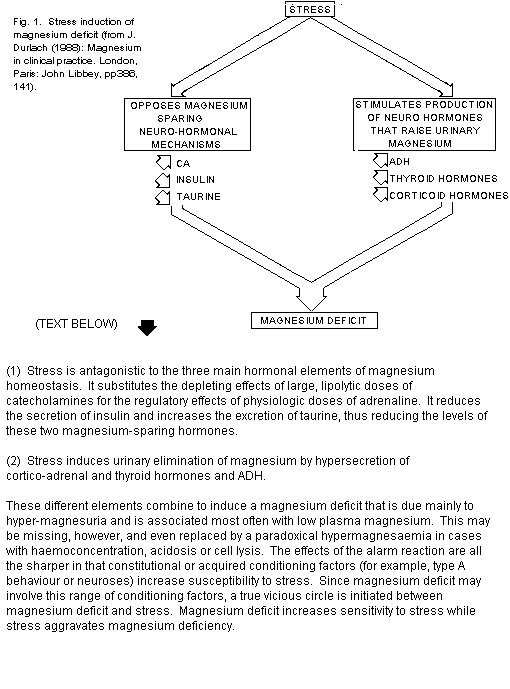
Conversely magnesium deficit creates a state of
hypersusceptibility to stress, even in cases of chronic marginal
deficit1,6. Thus magnesium deficit and stress
reinforce each other in a pathogenic vicious circle (Fig. 2, The
pathogenic "vicious circle": magnesium deficit-stress.)
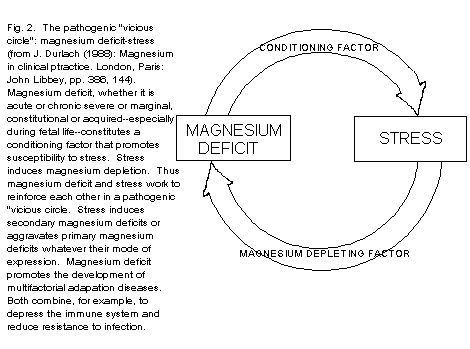
Constitutional conditioning factors
Constitutional conditioning factors may intervene in
individual differences in Mg requirements at several stages. In
the cell the efficiency of the enzyme activities may be variable,
requiring different levels of magnesium as cofactors.
Neurohormonal factors which either regulate or disturb magnesium
metabolism may be constitutionally more or less labile. Two
markers of these constitutional differences are currently
available. In people carrying the Bw35 allele,
erythrocyte magnesium--and to a lesser extent plasma
magnesium--are lower than in all the other HLA
groups7. The interindividual HLA typing differences in
magnesium metabolism reflect genetic differences in the activity
of the quinidine-sensitive Na2+/Mg2+
exchange8. Secondly, using a synchromatic Jenkins test
to determine behavioural type A or B, it has been shown that
classification as type A indicates predisposition to magnesium
deficit, probably because of an increased stress susceptibility
and a trend to high alcohol ingestion1.
Balance studies
I shall now discuss the evaluation of Mg RDA in developed
countries in relation to balance studies in healthy adults and
the respective values of other evaluations of magnesium
requirements.
Data from Mg balance studies
As early as 1964 9 and subsequently in updated
papers2,10-12, M.S. Seelig, analysing a large review
of balance studies in developed countries, concluded that the
minimum Mg intake required to maintain metabolic balance in
equilibrium was about 6 mg/kg body weight per day, that is to say
330 mg for a 55 kg woman and 420 mg for a 70 kg man. As early as
1968, our initial personal data13 and subsequent
updated reviews1,14 have been in good agreement with
this level of Mg intake to ensure zero or positive Mg balance
(Fig. 3, A comparative study in men and women of magnesium
balance...). It is appropriate to note that it is customary when
determining Mg balances to ignore not only the small
dermoskeletal and menstrual losses but also magnesium in sweat,
which can be a real accessory mechanism of deficit. Thus one
often obtains an exaggeratedly optimistic estimate of Mg
balance1,10.
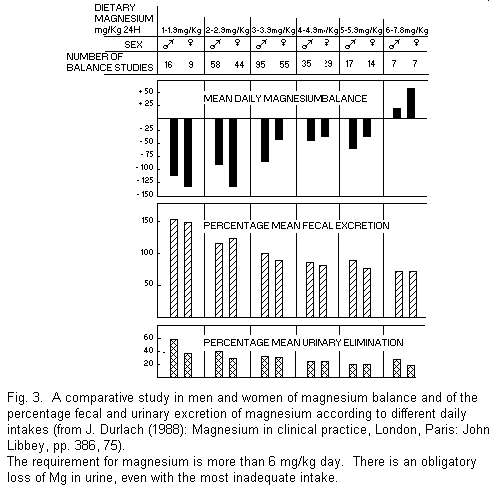
Significance of balance studies
It appears perfectly logical that an intake of a nutrient less
than the sum of the daily losses must at some time result in
deficiency and that such an intake must therefore be considered
inadequate. Nevertheless this assumption may be erroneous in
determining human requirements for minerals if considerable
adaptation of the pool size to the habitual intake is possible
(with concomitant conservation of good health, independently
evaluated)15. This is not usually the case with
magnesium.
Efficiency of the regulatory mechanism for Mg overload
Regulation of Mg metabolism against an overload is very
efficient. The renal reabsorptive mechanisms of Mg function at or
very near saturation. In healthy persons, compensating
hypermagnesuria represents an effective passive regulatory
mechanism for Mg overload. Positive Mg balance in healthy
subjects does not induce toxicity1 and negative Mg
balance does not arise from a compensatory mechanism of a Mg
overload1.
Lability of regulatory mechanisms for Mg deficit
The complex mechanisms which tend to compensate for an
insufficient magnesium intake are most often labile active
mechanisms. Their failures are well known, and in keeping with
narrow limits of adaptation to negative balances for an ion of
which the needs are high1. Positive balances, after an
insufficient intake of magnesium, indicate retention of the ion
to satisfy a thirst for magnesium created by Mg deficiency, the
consequences of which will therefore progressively be
alleviated1.
Importance of Mg turnover
Daily turnover of magnesium is particularly high. The ratio of
the Mg pool to total body Mg is about 8.1%, a ratio more than 15
times that of the corresponding selenium ratio, 30 times that of
calcium and 400 times that of iron. This very high turnover is
not really compatible with a compensatory decrease in the pool
size when the habitual intake is reduced. Conversely the high
need for exchangeable Mg for normal health induces a substantial
increase in the pool size during severe Mg deficit, which might
be a defence reaction1,16.
Thus the effective passive regulatory mechanism for Mg
overload, the lability of the active regulatory mechanisms for Mg
deficit, and the large need for exchangeable Mg are factors which
attribute special importance to Mg balance studies in determining
Mg intake sufficient to prevent negative magnesium balance and
magnesium deficiency.
Other evaluations of Mg RDA
In animal husbandry and in experimental nutrition--in
vitro or in vivo--a minimal intake to prevent
partial aspects of a mineral deficiency (growth, reproduction,
lactation) may be studied. But in human nutrition such studies of
functional requirement are not permissible for evident ethical
reasons. Nevertheless the evaluation of Mg RDA in terms of the
amount required to prevent hypomagnagnesaemia appears analogous
to these laboratory-based studies, and there is now much
experimental and clinical data showing that severe magnesium
deficit may exist without a decrease in
magnesaemia1,2,5,16. In the human being, Mg RDA must
be optimal for health, obviously insuring a normal degree of
storage and preventing the development of marginal primary
magnesium deficit.
Marginal primary Mg deficit
Overt and marginal deficiency and Mg requirements
Firstly, and most to blame for physicians' reluctance to
accept the possibility that Mg deficiency might be contributory
to medical problems, is the classical concept that the daily
requirement of a nutrient is the amount that prevents overt signs
and symptoms of deficiency. Acute and subacute experimental and
clinical data on Mg deficit have contributed greatly to knowledge
of the physiological and pathological importance of
Mg1,11,17. But it is the experimental clinical form of
marginal primary Mg deficit which is linked with marginal
intakes1,2,5,18.
Marginal primary Mg deficit in human beings
Neuromuscular hyperexicitability due to marginal primary
magnesium deficit accounts for its nonspecific symptomatology.
Latent tetany, with or without mitral valve prolapse, allergy or
pseudoallergy represent by far the most frequent expressions of
marginal magnesium deficiency.
Prevalence
Latent tetany due to primary magnesium deficit affects a large
proportion of the population (15 to 20%)1,18,24. This
large prevalence--established in Europe--is not surprising
because of the marginal nature of the magnesium intake. The
magnesium concept of the so-called "idiopathic mitral valve
prolapse" has important epidemiological implications: the high
prevalence of this disorder (5% of the population in the USA)
implies a three- to four-fold higher incidence of latent tetany
due to primary magnesium deficit since "idiopathic mitral vale
prolapse" occurs in only one third or one quarter of the cases.
Therefore, both in the USA and in Europe, latent tetany due to
primary magnesium deficit should affect a large part of the
population; such a similar prevalence would not be surprising
since the marginal nature of the magnesium intake is much the
same in the two continents23,24. Furthermore this
prevalence of marginal primary magnesium deficit seems consistent
with the estimation of nutrient deficiency using probability
analysis25,26 in populations where the mean Mg daily
intake is slightly above 4mg/kg day1-3,13,14,27-38
versus Mg RDA set at 6mg/kg day1-3.
Physiological oral Mg load test
Since the principal form of expression of marginal dietary
intake remains latent tetany due to magnesium deficit, it is
essential to seek out its manifestations and in general the
diagnosis relies on the magnesium oral loading test conducted
with physiological doses17-24. The dose of magnesium
to be administered is 5mg/kg day of a well absorbed salt for at
least one month, because of the high Mg turnover. At this
physiological dose level oral magnesium is totally devoid of the
pharmacodynamic effects of parenteral magnesium. Correction of
symptoms (clinical or paraclinical [tracings or biochemical
data], non-specific [other ions, enzymes] or specific [Mg
concentrations, usually in plasma, erythrocytes and daily urine,
and in lymphocytes when possible]) constitute the best proof that
the clinical pattern is due to an insufficient magnesium intake.
These variables should be checked after one month of
physiological oral magnesium supplementation. In forms of
magnesium deficit where depletion is due to dysregulation of the
factors which control magnesium metabolism, the response remains
partial after the one month physiological loading test; in these
cases the test should be followed by the best possible
investigation of the magnesium dysregulating
factors1,18-24.
Conclusions
Two types of further research appear necessary: long-term
studies of magnesium balance in adults consuming self-selected
diets in European countries, and magnesium intervention
trials.
New Mg balance studies in Europe
The importance of balance studies in determining RDA requires
new projects to be started in European countries. Food habits and
environmental factors may intervene in the estimation of the RDA.
The results of long-term studies of magnesium balance in adults
consuming self-selected diets are more significant than those
carried out in metabolic units, which may be biased by the
particular psychological profiles of volunteers and by the
different types of foods chosen, i.e. foods with
different phosphorus levels39,40. Self-selected diets
usually reveal negative magnesium balances4,27-40. In
order to update food composition tables it might be interesting
to compare the magnesium levels in raw, processed and cooked
samples of the diet with the data obtained from diet
histories41. These studies should involve various
socioeconomic groups and be followed by supplementation
studies.
Mg intervention trials
Intervention studies should be planned to determine whether
magnesium supplements given to at risk population groups will
reduce the incidence of the risk. The well-defined populations
should be those presenting with neuromuscular hyperexcitability
due to primary magnesium deficiency with or without "idopathic"
mitral valve prolapse, allergy or pseudoallergy. Therefore these
studies should deal mainly with the efficiency of magnesium
supplementation in latent tetany (or hyperventilation syndrome)
and in Barlow's disease. The same type of studies in populations
with cardiovascular disease or urolithiasis seems more hazardous
because of the multiplicity of their causes.
When choosing a magnesium supplement the energy intake should
be taken into consideration. Supplementation should be achieved
using a high magnesium density nutrient3,26,34,42,43
with the best possible bioavailability1,3. This
requirement is met by magnesium in water whether in its natural
form--from the tap or bottled--or in an artificial form by
addition of a soluble Mg salt to ordinary water1,3,44.
Supplementation of bread with magnesium has been
considered45, as well as specially formulated table
salts46,47. However these new salts have a triple
purpose since they increase both the Mg and K intake while
decreasing the Na intake, and they thus correspond to a more
complex intervention.
References
1. Durlach, J. (1988): Magnesium in clinical
practice, pp. 386. London, Paris: John Libbey.
2. Seelig, M.S. (1986): Nutritional status and requirements of
magnesium with consideration of individual differences and
prevention of cardiovascular disease. Magnesium-Bull.
8, 171-185.
3. Durlach, J., Bara, M. & Guiet-Bara, A. (1989):
Magnesium level in drinking water: its importance in
cardiovascular risk. In Magnesium in health and disease,
ed Y. Itokawa & J. Durlach pp. 173-182. London, Paris: John
Libbey.
4. Lakshmanan, F.L., Rao, R.B., Kim, W.W. & Kelsay, J.L.,
(1984): Magnesium intakes, balances and blood levels of adult
consuming self-selected diets. Am. J. Clin. Nutr.
40, 1380-1389.
5. Drueke, T., Gairard, A., Guegen, L., Hercberg, S. &
Mareschi, J.P. (1986): Minéraux en alimentation humaine.
Apports nutritionnels recomandés pour divers groupes
d'individus bien portants. Actualisation du calcium, du fer, du
phosphore et du magnésium. Cah. Nutr. Diet.
21, 339-356.
6. Heroux, O., Peter, D. & Heggtveit, A. (1977): Long term
effect of suboptimal dietary Mg on Mg and Ca contents of organs
on cold tolerance and lifespan, and its pathological consequences
in rats. J. Nutr. 107, 1640-1652.
7. Henrotte, J.G. (1989): Recent advances on genetic factors
regulating blood and tissue magnesium concentrations.
Relationships with stress and immunity. In Magnesium in
health and disease, ed Y. Itokawa & J. Durlach. London,
Paris: John Libbey.
8. Feray, J.C., Franck, G., Garay, R. & Henrotte, J.G.
(1989): Inter-individual differences in red cell Mg2+
contents are related to the activity of a
Na2+-Mg2+ exchanger. Possible relationship
with HLA-associated genetic factors (abstract). Magnesium
Res. 2, 124.
9. Seelig, M.S. (1964): The requirement of magnesium by the
normal adult. Summary and analysis of published data. Am. J.
Clin. Nutr. 14, 342-390.
10. Seelig, M.S. (1971): Human requirement of magnesium:
factors that increase needs. In 1st International Symposium
on Mg Deficit in Human Pathology,, Vol. 1, ed J. Durlach,
pp. 11-38. Vittel: SGEMV publ.
11. Seelig, M.S. (1980): Magnesium deficiency in the
pathogenesis of disease: early roots of cardiovascular, skeletal
and renal abnormalities, p. 488. New York: Plenum Press.
12. Seelig, M.S. (1981): Magnesium requirements in human
nutrition. Magnesium-Bull. 3,
26-47.
13. Amiot, D., Hioco, D. & Durlach, J. (1969):
Fréquence du déficit magnésique chez le
sujet normal et dans diverses ostéopathies. J. Med.
Bescançon 5., 371-378.
14. Durlach, J., Rayssiguier, Y. & Laguitton, A. (1980):
Le besoin en magnésium et son apport dans la ration.
Méd. Nutr. 16, 15-21.
15. Mertz, W. (1987): Use and misuse of balance studies.
J. Nutr. 117, 1811-1813.
16. Rousselet, F. & Durlach, J. (1971): Techniques
analytiques et explorations pratiques du métabolisme
magnésique en clinique humaine. In 1st International
Symposium on Mg Deficit in Human Pathology, Vol. 1, ed J.
Durlach, pp. 65-90. Vittel: SGEMV publ.
17. Shils, M.E. (1969): Experimental production of Mg
deficiency in man. In The pathogenesis and clinical
significance of Mg deficiency, ed E.B. Flink & J.E.
Jones, pp. 847-855. Ann. N.Y. Acad. Sci.
162.
18. Durlach, J. (1989): Magnesium: clinical forms of primary
magnesium deficit. Workshop on "Modern life styles, lower energy
intake and micronutrient status". Palma de Mallorca, 2-4 March
1989. ILSI Europe (in press).
19. Durlach, J. (1969): Spasmophilia and magnesium
deficit (Spasmophilie et déficit magnesique), pp 142.
Paris: Masson.
20. Durlach, J. (1976): Neurological manifestations of
magnesium imbalance. In Handbook of clinical neurology
ed P. Vinken & G.W. Bruyn, pp. 545-579. Amsterdam, N.Y.,
Oxford: North Holland.
21. Durlach, J. (1980): Clinical aspects of chronic magnesium
deficit. In Magnesium in health and disease, ed M.
Cantin & M.S. Seelig, pp. 883-909. New York, London: Spectrum
Press.
22. Durlach, J. (1985): Neurological disturbances due to
magnesium imbalance. In Metal ions in neurology and
psychiatry, ed S. Gabay, J. Harris & B.T. Ho, pp.
121-128. New York: A.R. Liss publ.
23. Durlach, J. & Durlach, V. (1986): Idiopathic mitral
valve prolapse and magnesium. State of the art.
Magnesium-Bull. 8, 156-159.
24. Durlach, J. (1988): Les rapports entre le magnésium
et le prolapsus de la valve mitrale. Revue Med.
Fonctionnelle 20: 121-172.
25. Anderson, G.H., Peterson, R.D. & Beaton, G.H. (1982):
Estimating nutrient deficiencies in a population from dietary
records: the use of probability analysis. Nutr. Res.
2, 409-416.
26. Harper, H.E. (1987): Evolution of Recommended Dietary
Allowances. New Directions? Annu. Rev. Nutr.
7, 509-537.
27. Greger, J.L., Gruner, S.M., Ethyre, G.M., Abernathy, R.P.
& Sickles, V. (1979): Dietary intake and nutritional status
in regard to magnesium of adolescent females. Nutr. Rep.
Int. 20, 235-244.
28. Huber, H.G., Disney, G.W. & Mason, R.L. (1981):
Urinary excretion and dietary intake of magnesium in girls.
Nutr. Rep. Int. 23, 127-134.
29. Tallas, P.G. (1981): Dietary intake of female college
students. Proc. Alaska Sci. Conf. 32,
14.
30. Marier, J.R. (1982): Quantitative factors regarding
magnesium status in the modern-day world. Magnesium
1, 3-15.
31. Morgan, K.J., Stampley, G.L., Zabik, M.E. & Fisher,
D.R. (1985): Magnesium and calcium dietary intakes of the US
population. J. Am. Coll. Nutr. 4,
195-206.
32. Morgan, K.J., & Stampley, G.L. (1985): Low magnesium
to calcium ratios in American self-selected diets. J. Am.
Coll. Nutr. 4, 344.
33. Chorazy, W. & Bliwert, K. (1985): Evaluation of
magnesium consumption in chosen population in Poland as a measure
of its proper concentration in a body. Magnesium-Bull.
7, 161.
34. Rouaud, C. (1985): Le magnésium. Information
Diététique Part 3 22-30.
35. Rouaud, C. (1986): Consommation alimentaire
d'étudiantes de la région parisienne: étude
particulière du magnesium. Méd. Nutr.
22, 295-300.
36. Guilland, J.C., Moreau, D., Boggio, V., Durlach, J. &
Klepping, J. (1986): Evaluation of dietary magnesium intake in
France. Magnesium-Bull. 8, 269.
37. Hill, A.D., Morris, E.R., Ellis, R., Moy, T. & Moser,
P.B. (1986): Mineral balance of adult men consuming self-selected
diets. Fed. Proc. 45, 375.
38. Desenclos, J.C., Rouaud, S., Soustre, Y., Berthier, A.M.,
Hercberg, S. & Dupin, H. (1988): Magnésium
hyperexcitabilité neuromusculaire et pression
artérelle: Etude dans une population de femmes
salariées de la region parisienne. Med. Nutr.
24, 279-284.
39. Suzuki, K., Uehara, M., Endo, Y. & Goto, S. (1988):
The effects of dietary magnesium levels on excess phosphate diet.
J. Japn. Soc. Magnesium Res. 7,
117-122.
40. Franz, K.B. (1989): Influence of phosphorus on intestinal
absorption of calcium and magnesium. In Magnesium in health
and disease. ed Y. Itokawa & J. Durlach. London, Paris:
John Libbey.
41. Riche, D. & Desenclos, J.C. (1985): Apports en
magnésium. Etude comparative de quelques tables de
composition. Med. Nutr. 21,
351-356.
42. Astier-Dumas, M., Gargominy, N. & Hoint, F. (1984):
Densité nutritionelle en magnésium: à propos
de quelques produits prêts à être
consommés. Méd. Nutr. 20,
397-399.
43. Riché, D. (1986): La couverture des besoins en
magnésium: un problème de densité
nutritionelle. Information Diététique,
Oart 4, 23-27.
44. Hese, A., Weber, A. & Miersch, W.D. (1988):
Magnesium-Substitution durch Minerali-vasser.
Therapie-Woche 38, 2510-2513.
45. Ranothra, G.S., Lee, C. & Gelroth, J.A. (1980):
Expanded cereal fortification: bioavailability and functionality
(flavor) of Mg in bread. J. Food Sci.
45, 915-917.
46. Pitkanen, H. (1982): Industrial possibilities to interfere
with the salt problem: dietary Na(K + Mg) ratio.
Magnesium 1, 298-303.
47. Angeli, G., Moretti, G.B., Bertone, V. & Amaglio, A.
(1988) Dieta iposodica e ipertensione arteriosa: effeto
favorevole sul colesterolo HDL di una nuova associazione
polisalina iposodica. Clin. Ter. 125,
417-424.
All articles by Dr. Durlach are copyrighted, and permission is
granted to Web users only to make single hard copies for personal
use. Additional reprints should be obtained from the originating
journals. Excerpts may be used by the media with attribution to
Dr. Durlach.
This page was first uploaded to The Magnesium Web Site on
December 23, 1995
http://www.mgwater.com/



